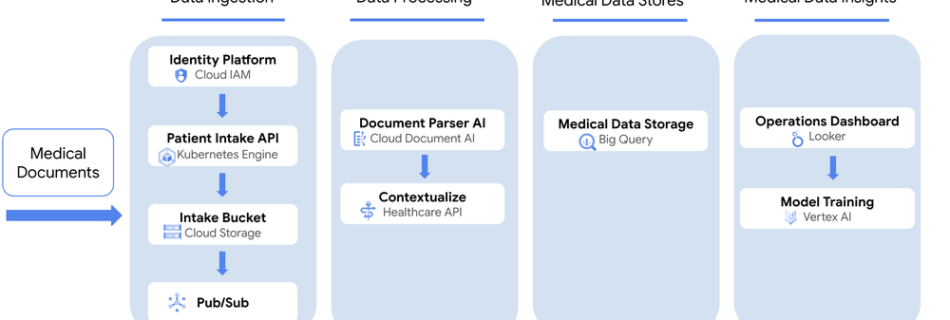
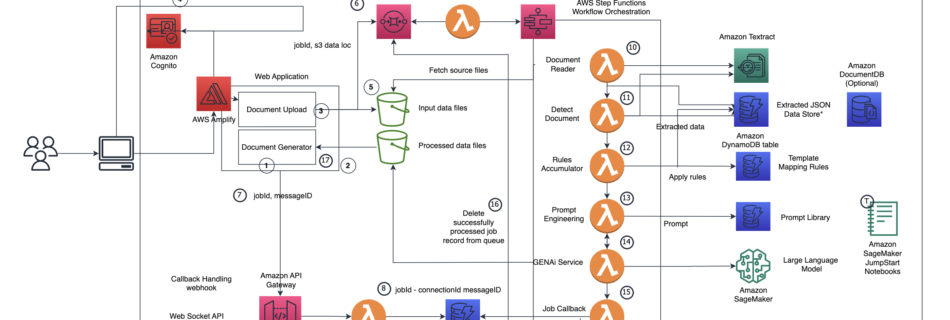
Medical Text Processing on Google Cloud
The FDA has a history of using real world evidence (RWE) as an integral component of the drug approval process. Moreover, RWE can mitigate the need for placebos in some clinical trials. The clinical records that make RWE evidence useful, however, often reside in unstructured formats, such as doctor’s notes, and must be “abstracted” into …